This occurs because as the liquid is heated, the force of attraction between. customary units) Specic Dynamic Kinematic Surface Modulus of Vapor Temperature Weight Density aViscosity Viscosity Tensionb Elasticity Pressure T Epv ( F) (lb/ft 3) (slug/ft3)(×105 lbs/ft2)(×105 ft2/s) (lb/ft) (10 lb f/in. This is especially true for the viscosity of water, for which relative viscosity data were first smoothed and then pinned to the recent value of 10.019☐.003 millipoises at 20☌ as reported by Swindells and Coe.įrom other empirical equations relating densities and energies of vaporization to temperature, equations are derived for expressing the activation energies for viscous flow as a function of temperature. The viscosity of most fluids decreases as its temperature increases and vice verse. 1862 Appendix C Physical Properties of Water Table C-2 Physical properties of water (U.S. As judged by the Gaussian criterion of fit, an expression suggested by the Eyring theory provides smoothed values for the viscosities of both water and mercury which are believed to be more reliable than those in common use. Working from the Navier-Stokes model of a compressible fluid one can show that the bulk viscosity makes a significant and measurable contribution to the. It was of further interest to apply the Arrhenius expression for the variation of viscosity with temperature to the data for both water and mercury and to compare the closeness of fit with that obtained by the use of the aforementioned expressions. Because water is frequently used as the calibrating fluid in viscometry, it was of interest to apply expressions, suggested by the theory of Eyring, for smoothing experimental data. For all of these mechanisms, the key underlying control on groundwater movement is the viscous resistance resulting from the interaction of the fluid with solid surfaces in the aquifer (grain edges or fracture walls).Smoothed values for the viscosities of water and mercury, as reported in the literature, show surprisingly large percentage deviations from the experimental data. Tortuosity is a measure of how far fluid must go to “circumnavigate” its way around particles: higher tortuosity indicates that water must go farther to get to its destination (a more tortuous path). But what controls their magnitude? The main factors are grain size and shape, sorting, porosity (degree of compaction or fracture aperture), particle orientation or alignment that affects the tortuosity of the flow path, and cementation. Prandtl number of water at 1, 10 and 100 bara (14.5, 1 psia), varying temperature given as ☌ or ☏: Back to top. 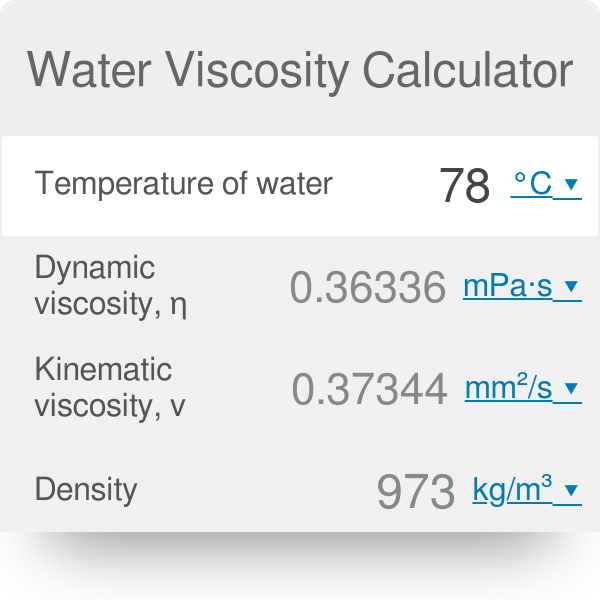
Prandtl number of water at 1 bara pressure, varying temperature given as ☌ or ☏: Back to top. 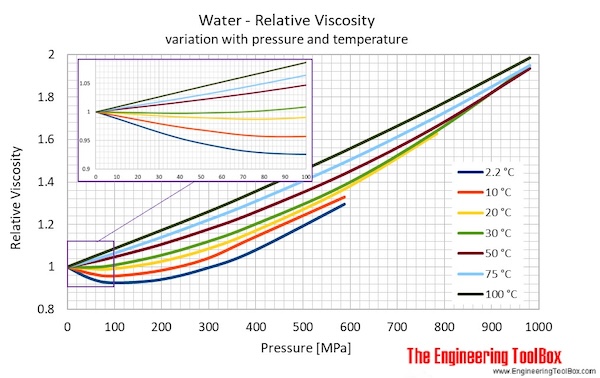
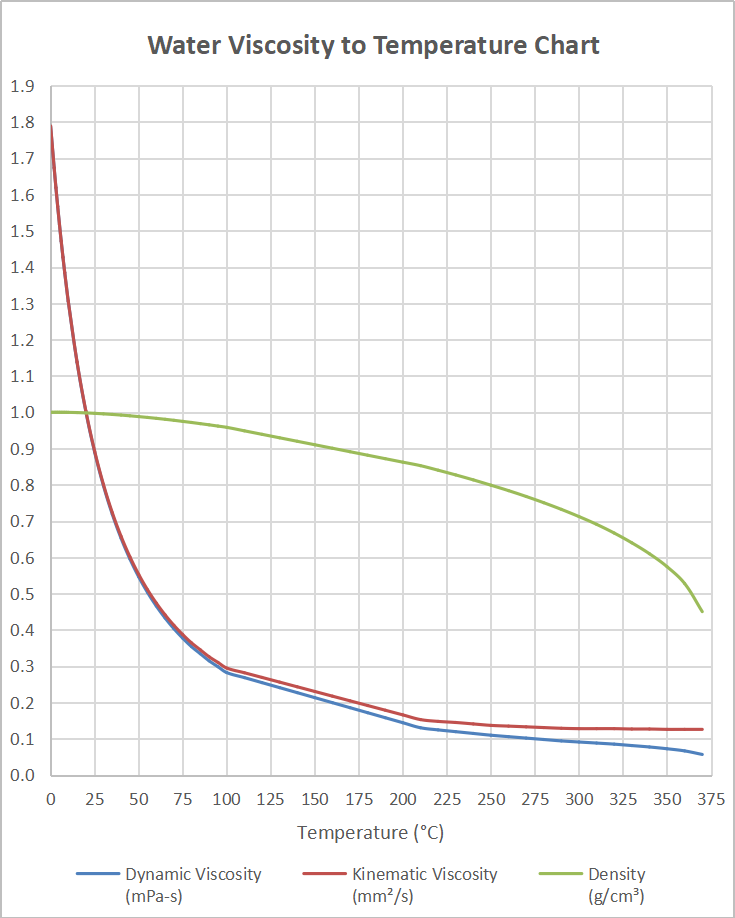
So…that’s how we define permeability and hydraulic conductivity. as well as Prandtl number of Air, Ammonia, Carbon dioxide, Methane, Nitrogen and Propane. The absolute or dynamic viscosity of water depends on the temperature as indicated below: 1 cP 0.01 poise 0.01 gram per cm second 0.001 Pascal second 1 milliPascal second 0.001 N.s/m2 Convert viscosity Water - Relative Viscosity vs. “thinner”), it will flow more easily through the aquifer. So even for the same aquifer, the hydraulic conductivity goes up if it is warmer! This makes some sense – if the water is less viscous (i.e. Water viscosity is independent of fluid pressure except for very high values larger than 80 MPa, yet it is strongly dependent on temperature (Likhachev, 2003). Around room temperature, the viscosity of water decreases by 3 per degree Celcius oils by about 5, and bitumen by 15 or more. It is also important in considering the effects of temperature, because water viscosity decreases with increasing temperature: it’s less than half as viscous at 90° than at 32° F. 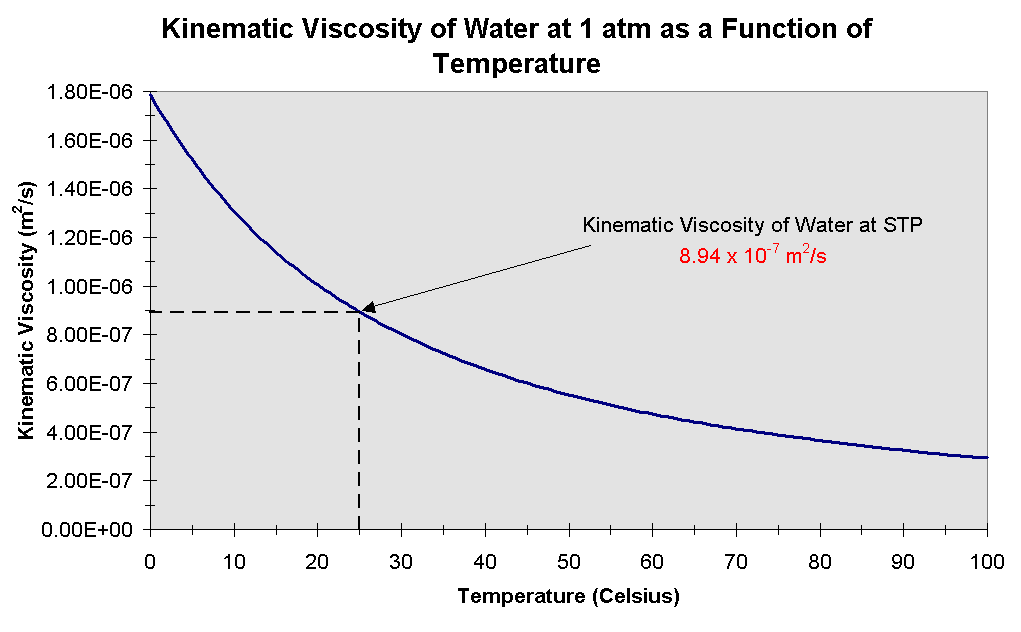
water – whether you are thinking about an oil reservoir or contamination of groundwater by a gasoline spill). This is important for comparing different fluids (say, oil vs. The water molecules have less kinetic energy to move at lower temperatures meaning the water does. More viscous fluids will flow more slowly through the same rock than less viscous ones. The water viscosity decreases as the temperature increases. More specifically, it is the viscosity and density of the fluid that matter. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
